Our Portfolio
Prolynx is addressing a fundamental challenge facing chronic care in obesity and metabolic disease:
How to maintain consistent drug exposure over extended periods in a way that reduces dosing burden and supports sustained treatment over time.
Built into the portfolio
Tunable release profiles
The Prolynx delivery approach enables adjustable release kinetics to support different exposure profiles and dosing intervals. This tunability allows dosing to be engineered around specific pharmacological objectives without redesigning the active drug itself.
Predictable pharmacokinetics
Controlled release is designed to support highly predictable and reproducible pharmacokinetic profiles across long dosing intervals. Maintaining stable exposure and tightly controlled plasma concentrations offers efficacy and tolerability benefits while avoiding pitfalls such as burst release, which is common with other half-life extension approaches.
Designed for long-term exposure
Conjugates are engineered to maintain therapeutically relevant drug levels over prolonged periods from a single administration. This design prioritizes sustained exposure and stability over time rather than short-lived peak effects.
Compatible with combination strategies
Prolynx’s highly modular technologies are designed to enable pharmacokinetically matched combinations of peptide-based therapies. By harmonizing the release profiles of multiple compounds, Prolynx enables long-acting combination approaches, regardless of each compound’s intrinsic pharmacokinetics.
Lead Program
PLX-821 (semaglutide) is Prolynx’s lead ultra-long-acting therapeutic candidate for obesity and type 2 diabetes, built on the company’s long-acting delivery architecture.
.png)
Broader Pipeline
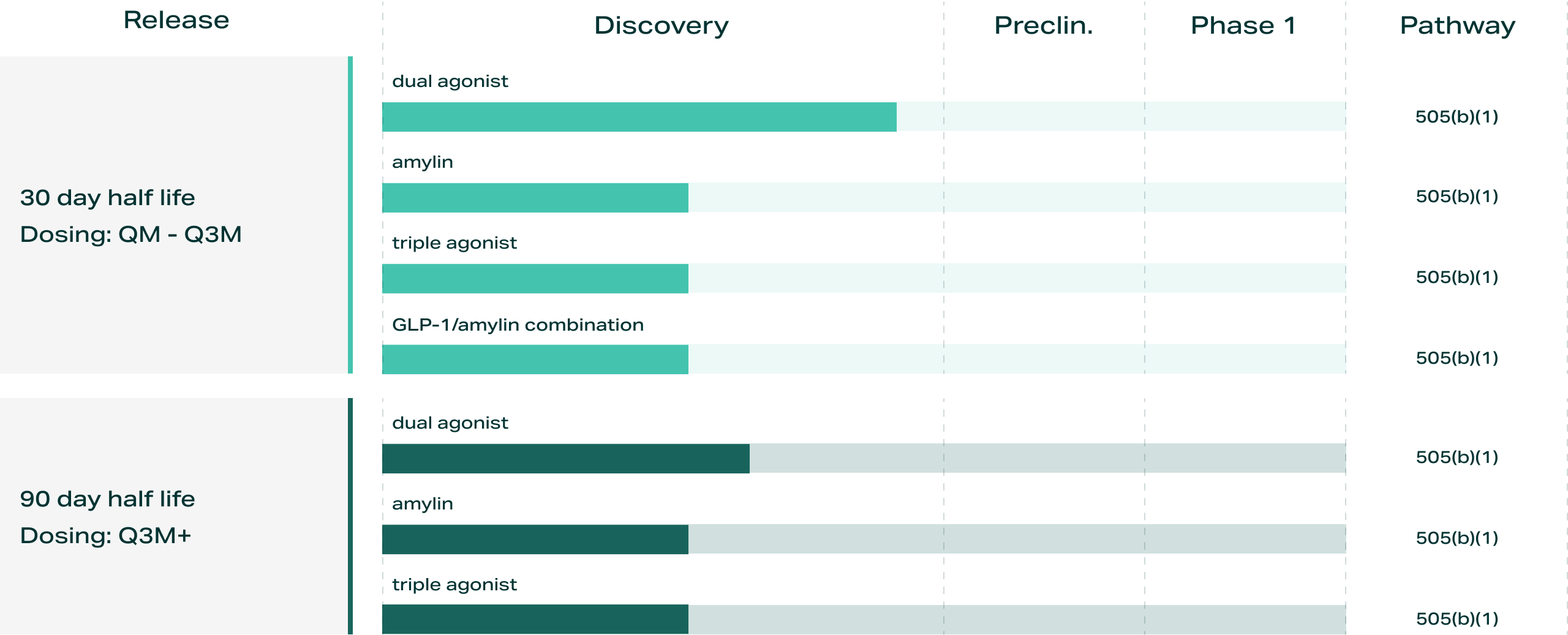
We are advancing a pipeline of ultra-long-acting programs across multiple metabolic mechanisms, including dual and triple agonists, amylin-based therapies, and combination approaches, all designed to leverage the same extra long acting framework with the potential to enable extended dosing intervals of up to 90 days and pharmacokinetically matched combinations.

A modular platform for ultra-long-acting therapies
Prolynx’s platform uses a hydrogel-based microsphere system with tunable release linkers to control drug release over time. The approach is designed to support ultra-long-acting exposure with consistent pharmacokinetics from a single administration and can be applied across multiple therapeutic programs, mechanisms and combination strategies.

Microsphere gel
A degradable tetra-PEG–based hydrogel microsphere that forms a stable subcutaneous depot for slow drug release over time.
Tunable release linker
A proprietary, tunable linker chemistry that governs release kinetics and enables highly predictable exposure without burst release.
Active drug
The active therapeutic payload is covalently bound to the carrier and released gradually from the subcutaneous depot in its native form.
TEAM
Core Team
Chris Boulton
CEO

Chris Boulton is Chief Executive Officer of Prolynx, overseeing portfolio advancement and organizational scale as the company advances its proprietary linker technology for ultra-long-acting therapeutics. Previously, he held senior commercial leadership roles across global pharmaceutical and biotech organizations, where he led product launches and commercial strategy across obesity, metabolic, and specialty therapeutics.
Earlier in his career, Chris built and led global sales and marketing organizations and advised emerging biotech companies on commercial scale-up and market entry.
Alexander Steiner
Chief Business Officer

Alex Steiner is Chief Business Officer and Head of R&D Operations at Prolynx, where he supports corporate strategy for the company’s ultra-long-acting anti-obesity portfolio. Previously, he served as Vice President and Head of Business Development at Prellis Biologics, where he helped guide the company’s evolution from a technology platform to a pipeline-focused drug development company.
Earlier in his career, Alex was an early employee at Sutro Biopharma, where he supported platform build-out and major pharma collaborations. He holds an MBA from The Wharton School and a B.S. and M.S. in Chemical Engineering from Stanford University.
Daniel Santi, M.D., PH.D.
(Co-Founder, Chief Technical & Scientific Officer)

Daniel Santi, M.D., Ph.D., is a co-founder of Prolynx and serves as Chief Technical and Scientific Officer, where he provides scientific leadership for the company’s sustained drug delivery platform. Previously, he co-founded and led multiple biotechnology companies, including Kosan Biosciences, where he served as CEO and Chairman through its public offering and subsequent acquisition by Bristol Myers Squibb.
Earlier in his career, Dr. Santi held faculty and translational leadership roles at the University of California, San Francisco, including Director of Translational Research at QB3, where he built industry-facing research and partnership programs. He has authored over 300 scientific publications and is a co-inventor on more than 50 U.S. patents.
Gary Ashley, Ph.D.
Co-Founder

Dr. Ashley is a co-founder of Prolynx and has spent more than a decade refining the chemistry underlying the company’s ultra-long-acting half-life extension technology. Previously, he served as Assistant Professor of Chemistry at Northwestern University and held senior chemistry and research leadership roles at Parnassus Pharmaceuticals and Kosan Biosciences, including Executive Director of Chemistry and Vice President of Exploratory Research.
Earlier in his career, he led drug discovery and exploratory research efforts across academic and industry settings, contributing to the advancement of multiple therapeutic programs. He holds a B.S. in Chemistry from MIT and a Ph.D. in Organic Chemistry from the University of California, Berkeley, and is a licensed patent agent with over 70 scientific publications and more than 50 issued U.S. patents
Christian Weyer, M.D., M.A.S.
Chief Medical Officer

Christian Weyer, M.D., M.A.S., is Chief Medical Officer at Prolynx, where he leads clinical and medical strategy for the company’s ultra-long-acting therapeutic portfolio. Previously, he held executive officer roles across private and public biopharmaceutical companies, including Escient Pharmaceuticals, Intercept Pharmaceuticals, Fate Therapeutics, and Amylin Pharmaceuticals, where he led R&D and clinical development efforts through acquisition and global partnerships.
He received his M.D. and clinical training in metabolic disorders at the University of Düsseldorf and holds a post-doctoral master’s degree in advanced clinical research from the University of California, San Diego.
Board of Directors
Chris Boulton is Chief Executive Officer of Prolynx, overseeing portfolio advancement and organizational scale as the company advances its proprietary linker technology for ultra-long-acting therapeutics. Previously, he held senior commercial leadership roles across global pharmaceutical and biotech organizations, where he led product launches and commercial strategy across obesity, metabolic, and specialty therapeutics.
Earlier in his career, Chris built and led global sales and marketing organizations and advised emerging biotech companies on commercial scale-up and market entry.
Daniel Santi, M.D., Ph.D., is a co-founder of Prolynx and serves as Chief Technical and Scientific Officer, where he provides scientific leadership for the company’s sustained drug delivery platform. Previously, he co-founded and led multiple biotechnology companies, including Kosan Biosciences, where he served as CEO and Chairman through its public offering and subsequent acquisition by Bristol Myers Squibb.
Earlier in his career, Dr. Santi held faculty and translational leadership roles at the University of California, San Francisco, including Director of Translational Research at QB3, where he built industry-facing research and partnership programs. He has authored over 300 scientific publications and is a co-inventor on more than 50 U.S. patents.
Kush Parmar, M.D., Ph.D. is Managing Partner at 5AM Ventures, where he leads investments in early-stage life science companies and works closely with founders on company strategy, leadership, and growth. Since joining the firm in 2010, he has helped build and scale multiple biotechnology and pharmaceutical companies.
Earlier in his career, Dr. Parmar served as Founding CEO and Chairman of Homology Medicines and has held numerous board and leadership roles across the biotech sector, including ProLynx, Entrada Therapeutics, Ensoma, and Audentes Therapeutics. He is a Kauffman Fellow and actively contributes to academic and scientific advisory councils.
Fred Cohen, M.D., D.Phil. is Co-Founder and Chairman of Monograph Capital Partners and Co-Founder and Senior Managing Director at Vida Ventures, where he focuses on building and supporting life science and biotechnology companies.
Earlier in his career, Dr. Cohen was a Partner and founder of TPG Biotechnology and held senior academic and clinical leadership roles at UCSF, including Chief of the Division of Endocrinology and Metabolism. He is a member of the National Academy of Medicine and the American Academy of Arts and Sciences.
Peter A. Thompson, M.D. is a General Partner at OrbiMed, where he leads investments and serves on the boards of multiple life science and biotechnology companies. He brings over 25 years of industry experience, including executive roles at Becton Dickinson and Chiron.
Earlier in his career, Dr. Thompson co-founded Trubion Pharmaceuticals and served as CEO of several biotechnology companies. He is an Affiliate Professor of Neurosurgery at the University of Washington and holds degrees in Molecular Biology, Mathematics, and Medicine from Brown University.
Lorem ipsum dolor sit amet consectetur. Ut in et pellentesque amet. Phasellus natoque quis lectus at nunc pellentesque maecenas vivamus odio. Ac morbi senectus diam neque. Venenatis tristique felis proin in hac. Malesuada elementum aliquam porttitor tellus non amet gravida. Magna nunc dictum praesent sed volutpat orci. Aliquam ullamcorper augue.
Lorem ipsum dolor sit amet consectetur. Arcu fringilla euismod vel hac. Quis posuere scelerisque libero enim. Justo gravida tristique ac aliquam in praesent vitae morbi. Imperdiet lectus iaculis blandit morbi non nunc. Aliquam orci neque ut laoreet. Blandit consectetur malesuada nunc est nisl mi. Fringilla egestas fames libero bibendum risus arcu eu ullamcorper.
NEWS








